Blog
How to Calculate Battery Run Time: The Complete Guide (2026)
2026-05-09 | Calvin
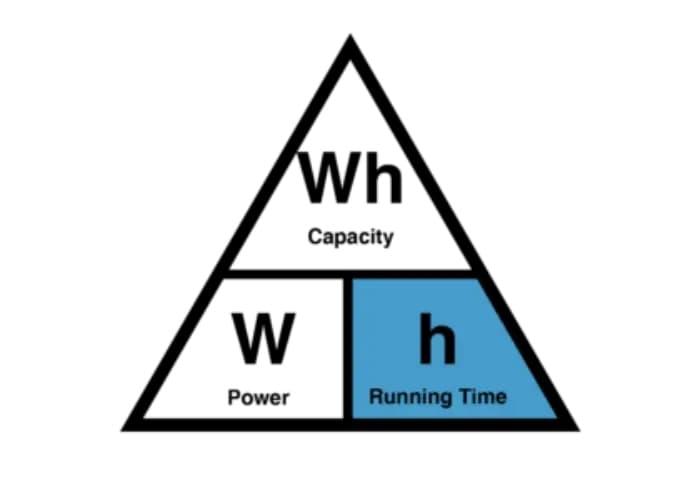
Table of Contents
- What Battery Run Time Really Means
- The 3 Core Formulas
- Step-by-Step Walkthrough
- 8 Real-World Examples
- Peukert's Law (High-Current Loads)
- Chemistry Comparison Table
- Factors That Reduce Real Runtime
- Parallel & Series Battery Configurations
- Troubleshooting: Why It Lasts Less
- FAQ
Key Takeaways
- The basic formula is Runtime (h) = Capacity (mAh) ÷ Current (mA) — but real-world runtime is typically 10–40% lower.
- Always convert to Watt-hours (Wh) when the device load is rated in watts — it avoids voltage mismatch errors.
- Apply Depth of Discharge (DoD) and system efficiency (η) multipliers for accurate real-world estimates.
- At high discharge rates, use Peukert's Law — it explains why a 100Ah battery may only deliver 70–80Ah under heavy load.
- LiFePO4 batteries allow 80–95% DoD vs. ~50% for lead-acid — delivering roughly double the usable runtime per Ah of rated capacity.
- Parallel battery configurations increase capacity (longer runtime); series configurations increase voltage (not runtime).
1. What Battery Run Time Really Means
Battery run time is the duration a battery can continuously power a load before reaching its minimum safe discharge voltage. It is expressed in hours and depends on two variables: how much usable energy is stored in the battery, and how fast the load is consuming that energy.
Two common points of confusion are worth clearing up immediately:
mAh is not the same as runtime. A 5,000 mAh battery does not automatically last 5 hours. The current draw determines whether it lasts 2 hours or 50 hours. mAh is just stored charge — not time.
Rated capacity ≠ usable capacity. A 100Ah lead-acid battery may only safely deliver 50Ah before damaging the cells. A 100Ah LiFePO4 battery can deliver 80–95Ah. Battery chemistry fundamentally changes how much of the rated number you can actually use.
Quick Reference Runtime shrinks when: discharge rate is high, temperature is extreme, the battery is aged, the load is variable (not constant), or an inverter/converter is in the circuit. We'll quantify each of these below.
2. The 3 Core Battery Run Time Formulas
Formula 1 — Current-Based (mAh ÷ mA)
Use this when your device's current draw is known and relatively constant — for example, a sensor, LED system, or low-power IoT device.
Method 1 · Current-Based
Runtime (h) = Battery Capacity (mAh) ÷ Load Current (mA)
Example: 4,000 mAh ÷ 500 mA = 8 hours (theoretical maximum)
Formula 2 — Power-Based (Wh ÷ W)
Use this when the device is rated in watts — inverters, laptops, appliances, power tools. This method is more accurate for AC loads and systems with voltage conversion.
Method 2 · Power-Based
Runtime (h) = (mAh × V ÷ 1000) ÷ Load Power (W)
Step 1: Convert mAh to Wh → Wh = mAh × Voltage ÷ 1000
Step 2: Divide → Runtime = Wh ÷ Watts
Formula 3 — Practical Estimate (Recommended)
This is the formula you should use for any real-world system design. It adds two critical correction factors: Depth of Discharge (DoD) and system efficiency (η).
Method 3 · Practical Estimate (Best Accuracy)
Runtime ≈ [(mAh × V ÷ 1000) ÷ W] × DoD × η
DoD = Depth of Discharge (usable fraction of capacity)
→ LiFePO4: 0.80–0.95 | Li-ion: 0.80–0.90 | Lead-Acid: 0.45–0.55
η = System efficiency (DC-DC converter, inverter, BMS losses)
→ Inverter: 0.85–0.92 | DC-DC step-down: 0.88–0.95 | Direct DC: 0.95–0.99
Unit Conversion Reference
mAh → Ah
Ah = mAh ÷ 1,000
3,000 mAh = 3 Ah
Ah → Wh
Wh = Ah × Voltage
3 Ah × 3.7V = 11.1 Wh
W = V × A
Amps = Watts ÷ Volts
10W ÷ 5V = 2A (2,000 mA)
Wh → Runtime
h = Wh ÷ Watts
50 Wh ÷ 10 W = 5 hours
3. Step-by-Step Walkthrough
Follow these five steps for any battery runtime calculation:
1 Identify Battery Capacity
Find the mAh or Ah rating on the battery label or datasheet. If rated in Ah, multiply by 1,000 to get mAh. For battery packs, check if the rating is per-cell or for the full pack.
2 Determine Load Demand
Check the device spec sheet. If rated in mA, use Formula 1. If rated in watts, use Formula 2. If the load is variable (e.g., a motor that spikes), use the average or RMS power draw, not the peak.
3 Convert to Watt-Hours
Multiply Ah × Voltage = Wh. This is the most reliable unit for cross-system calculations, especially when the battery voltage differs from the load voltage (e.g., 48V battery powering 12V equipment).
4 Apply DoD and Efficiency
Multiply theoretical runtime by your DoD and efficiency factors. For a LiFePO4 pack powering a quality inverter: DoD = 0.90, η = 0.90. This gives you an 81% multiplier — your real runtime is about 81% of theoretical.
5 Add Temperature Correction (if needed)
In cold environments (below 0°C / 32°F), subtract an additional 10–30% depending on chemistry. Lead-acid is most affected; LiFePO4 is most cold-resistant among common chemistries.
4. Eight Real-World Calculation Examples
Example 1 Flashlight / LED Light
Battery: 1,000 mAh, 1.5V
Load: 50 mA constant
1,000 ÷ 50 = 20h theoretical
× 0.80 DoD = 16h realistic
≈ 16 hours
Example 2 Smartphone
Battery: 4,500 mAh, 3.85V
Avg draw: ~400 mA
4,500 ÷ 400 = 11.25h theoretical
× 0.85 DoD = ~9.6h
≈ 9–10 hours
Example 3 Laptop
Battery: 56 Wh (built-in)
Load: 25W avg (light use)
56 ÷ 25 = 2.24h theoretical
× 0.88 DoD × 0.93 η = ~1.83h
at 45W load: 56 ÷ 45 × 0.82 = ~1.02h
≈ 1–2 hours (load-dependent)
Example 4 Power Tool
Battery: 5,000 mAh, 18V → 90 Wh
Motor load: 400W peak, 150W avg
90 Wh ÷ 150W = 0.6h (36 min)
× 0.85 DoD × 0.93 η = ~28 min avg
≈ 25–35 min continuous
Example 5 RV / Camper Van
Battery: 200Ah LiFePO4 @ 12V → 2,400 Wh
Load: 150W avg (fridge + lights + USB)
2,400 ÷ 150 = 16h theoretical
× 0.90 DoD × 0.93 η = ~13.4h
≈ 13 hours overnight
Example 6 Solar Off-Grid Inverter
Battery: 100Ah @ 12V → 1,200 Wh
AC Load: 400W, inverter η = 0.90
1,200 × 0.80 DoD × 0.90 η = 864 Wh usable
864 ÷ 400W = 2.16 hours
≈ 2.2 hours
Example 7 Racing Drone
LiPo: 1,500 mAh, 14.8V (4S), 70A avg draw
1,500 mAh ÷ 70,000 mA = 0.021h
= ~1.3 min at full throttle
At 40% avg throttle (28A): ~3.2 min
≈ 3–5 min flight time
Example 8 Medical Device (Portable Ventilator)
Battery: 28.8 Wh (internal), 7.4V
Continuous load: 18W
28.8 ÷ 18 = 1.6h theoretical
× 0.85 DoD × 0.95 η = ~1.29h
≈ 1.25 hours (critical — verify with OEM)
⚠ Important for Medical & Safety-Critical Devices
Always use the manufacturer's published runtime specifications for medical devices, emergency lighting, and life-safety systems. Calculated estimates are engineering approximations and should not substitute for certified operational testing.
5. Peukert's Law: High-Current Runtime Accuracy
The formulas above assume the battery delivers its full rated capacity regardless of how fast you draw from it. In reality, this is only true at low discharge rates. At high discharge rates, internal resistance causes heat and voltage sag, and the battery delivers less total energy than its rating suggests.
This relationship is described by Peukert's Law, published in 1897 and still used in battery engineering today. It is most significant for lead-acid batteries and matters less for modern lithium chemistries.
Peukert's Law — Capacity-Corrected Formula
T = (C / In) × Cn-1
T = Corrected runtime (hours)
C = Rated capacity at the reference discharge rate (Ah)
I = Actual discharge current (A)
n = Peukert exponent
→ Lead-acid: n = 1.1–1.3 (significant effect)
→ Li-ion / LiFePO4: n ≈ 1.01–1.05 (minimal effect — one reason lithium is preferred for high-current applications)
Practical Example of Peukert's Effect
A 100Ah lead-acid battery is rated at C/20 (5A discharge over 20 hours). If you discharge it at 50A instead:
Peukert Calculation
Effective capacity = 100 × (5 / 50)1.2 - 1 = 100 × 0.10.2 ≈ 63 Ah
The battery only delivers ~63Ah — not 100Ah — at this discharge rate. Runtime = 63 ÷ 50 = 1.26 hours, not the 2 hours Formula 1 would predict.
When to Apply Peukert's Law
Apply it when your discharge rate exceeds C/5 (i.e., discharging a 100Ah battery faster than 20A). For lead-acid in inverter/solar applications, this is common and explains why field results often fall well below calculator estimates.
6. Battery Chemistry Runtime Comparison
The same Ah capacity delivers dramatically different real-world runtime depending on battery chemistry. This table compares the four most common chemistries for a 100Ah battery pack powering a 100W load at nominal voltage.
| Chemistry | Nominal Voltage | Typical DoD | Peukert (n) | Usable Wh (100Ah) | Runtime at 100W | Cycle Life |
|---|---|---|---|---|---|---|
| LiFePO4 | 12.8V | 80–95% | ~1.02 | 1,075–1,216 Wh | ~10.8–12.2h | 2,000–6,000+ |
| Li-ion (NMC) | 14.4V | 80–90% | ~1.03 | 1,152–1,296 Wh | ~11.5–13.0h | 500–1,500 |
| Lead-Acid (AGM) | 12V | 45–55% | 1.1–1.3 | 540–660 Wh | ~5.4–6.6h | 300–600 |
| NiMH | 12V | 60–75% | ~1.05 | 720–900 Wh | ~7.2–9.0h | 500–1,000 |
* Calculations assume 100Ah rated capacity, nominal voltage, 93% system efficiency, 10°C–25°C operating temperature. Actual results vary with load profile and battery age.
LiFePO4 delivers roughly 1.8–2× the usable runtime of an equivalent lead-acid battery — not because it stores more energy per Ah, but because you can use a far higher percentage of its rated capacity on every cycle without damaging the cells.
7. Factors That Reduce Real Battery Runtime
Understanding what shrinks runtime in practice allows you to design systems that perform closer to theoretical limits:
| Factor | Typical Runtime Impact | Notes |
|---|---|---|
| Temperature (below 0°C) | −10% to −30% | Li-ion loses capacity faster than LiFePO4 in cold; lead-acid is most affected |
| High discharge rate (C-rate) | −5% to −25% | More significant for lead-acid; lithium is largely stable up to 1C–2C |
| Battery age / capacity fade | −5% to −30% | Most batteries retain ≥80% capacity at rated end-of-life cycle count |
| Inverter / converter losses | −7% to −15% | Budget-grade inverters can drop to 80–85% efficiency under partial load |
| Wiring / connection resistance | −1% to −5% | Use appropriate cable gauge and clean terminals to minimize losses |
| Self-discharge (stored battery) | ~1–5% per month | LiFePO4 self-discharges ~2–3% monthly; lead-acid ~5–15% |
| Partial state of charge operation | Variable | Lead-acid suffers sulfation when repeatedly not fully charged |
Real-World Planning Rule of Thumb
For system design purposes, plan for approximately 70–80% of theoretical runtime under normal conditions. In demanding environments (cold temperatures, high discharge rates, aging battery), budget for 55–65% of theoretical.
8. Parallel & Series Battery Configurations
When a single battery doesn't provide enough runtime or voltage, batteries are combined. The configuration determines whether you get more runtime or higher voltage.
Parallel Configuration (More Runtime)
Connect positive terminals together and negative terminals together. Voltage stays the same; capacity adds up.
Parallel: 2× 100Ah batteries @ 12V
Total = 200Ah @ 12V = 2,400 Wh
Runtime doubles. Use identical batteries of the same age and state of charge to avoid imbalance current flow between cells.
Series Configuration (Higher Voltage)
Connect positive of battery 1 to negative of battery 2. Voltage adds; capacity stays the same as a single battery.
Series: 2× 100Ah batteries @ 12V
Total = 100Ah @ 24V = 2,400 Wh
Same total Wh — same runtime at the same watt load. Series is used when your inverter or motor requires higher voltage, not to extend runtime.
Key Insight
Both a parallel pair and a series pair of 100Ah 12V batteries contain the same 2,400 Wh of total energy. Runtime at a given watt load is identical — the difference is voltage level, not duration.
9. Troubleshooting: Why Is My Battery Dying Faster Than Calculated?
You're operating in cold temperatures
Below 10°C, lithium battery capacity drops noticeably. Below 0°C, it can fall 20–30%. Lead-acid is even more sensitive — a fully charged lead-acid battery at −18°C may only deliver 40% of its rated capacity.
The battery is aged or degraded
Battery capacity naturally declines with use. A Li-ion battery that has completed 80% of its rated cycle life may only hold 80% of its original capacity. Check State of Health (SoH) with a battery analyzer if runtime has declined significantly from new.
Your load is higher than you measured
Motors, compressors, and inverters have startup surges (inrush current) that can be 3–5× the running current. Variable loads — like a fridge that cycles on and off — are best measured with a clamp meter over an extended period, not just at one instant.
Inverter or converter efficiency is lower than assumed
Inverter efficiency curves are non-linear. Many inverters are most efficient at 50–75% of rated load and drop to 80–85% efficiency at very light loads (<10% of rated). If your load is much smaller than the inverter's rating, efficiency losses compound.
Your battery's DoD limit is more conservative than you assumed
Many Battery Management Systems (BMS) cut off at 20% State of Charge (SoC) — meaning usable DoD is only 80%, not 100%. Some budget systems cut off at 30%, leaving only 70% usable. Check your BMS low-voltage cutoff setting.
The battery was never fully charged
A charger with too low a voltage setpoint will leave the battery partially charged. For a 12V LiFePO4 battery, full charge requires ~14.6V at the battery terminals. Voltage drop across long wiring runs can prevent this without proper charge compensation.
10. Frequently Asked Questions
How do I calculate battery run time from mAh and watts?
Convert mAh to Wh first: Wh = mAh × Voltage ÷ 1,000. Then divide by your load in watts: Runtime = Wh ÷ W. For real-world accuracy, multiply by DoD (typically 0.80–0.90 for lithium) and system efficiency (0.85–0.95). Example: a 20,000 mAh / 3.7V power bank powering a 10W device — theoretical Wh = 74Wh. Runtime = 74 ÷ 10 × 0.85 ≈ 6.3 hours.
How long will a 100Ah battery last running a 400W appliance?
At 12V: 100Ah × 12V = 1,200 Wh. Theoretical: 1,200 ÷ 400 = 3 hours. With LiFePO4 (DoD 0.90) and a 90% efficient inverter: 1,200 × 0.90 × 0.90 = 972 Wh usable. Runtime = 972 ÷ 400 = 2.43 hours. With lead-acid (DoD 0.50): 1,200 × 0.50 × 0.88 = 528 Wh. Runtime = 528 ÷ 400 = 1.32 hours. Chemistry matters enormously for this calculation.
Does cold weather really reduce battery run time?
Yes, significantly. Lithium-ion and LiFePO4 batteries can lose 10–20% capacity at 0°C and up to 30–40% at −20°C, depending on the discharge rate. Lead-acid batteries are more severely affected — some lose 50% capacity at −18°C. For outdoor or cold-climate applications, derate your expected runtime accordingly and consider insulating the battery enclosure.
What is the difference between battery run time and battery cycle life?
Run time is how long the battery lasts on a single charge — measured in hours. Cycle life is how many charge/discharge cycles the battery can complete before its capacity falls to 80% of the original rating — measured in cycles or years. A battery with long run time doesn't necessarily have a long cycle life, and vice versa. LiFePO4 excels at both; lead-acid is mediocre at both but low cost upfront.
What does C-rate mean and how does it affect runtime?
C-rate describes the discharge current relative to battery capacity. A 1C rate on a 100Ah battery is 100A discharge — it would theoretically last 1 hour. 0.5C = 50A = 2 hours. 2C = 200A = 30 minutes. At higher C-rates, Peukert's effect means you actually get less total energy out — a 100Ah lead-acid battery at 2C may only deliver 70–75Ah total. Lithium batteries are far less affected by C-rate, which is why they dominate high-power applications.
Can I extend my battery run time without buying a bigger battery?
Yes — several strategies: (1) Reduce your load. Switching from a 400W inverter load to a 200W load roughly doubles runtime. (2) Upgrade from lead-acid to LiFePO4 — you'll access nearly twice the usable capacity from the same Ah rating. (3) Keep the battery warm in cold environments. (4) Upgrade to a higher-efficiency inverter. (5) Check your wiring and connections — loose or undersized cables waste energy as heat. (6) Reduce background loads — parasitic draws from BMS, monitoring systems, and standby equipment add up over time.
- Next:26650 vs 18650 Lithium Batteries: A Comprehensive Comparison
- Previous:Spot Welding vs. Soldering Lithium Batteries: Which Method Should You Use?
Contact Details
Lithium LiFePO4 Batteries and Lithium LiFePO4 Cells Supplier - LiFePO4 Battery Shop
Contact Person: Miss. Elena Wang
WhatsApp : +8615263269227
Skype : +8615263269227
WeChat :15263269227
Email : info@lifepo4batteryshop.com
All Products
- TC Elcon Charger (0)
- Headway Lithium Battery (6)
- Blade Battery (10)
- Semi Solid State Battery (6)
- A123 Battery (6)
- Sinopoly Battery (7)
- GBS Battery (16)
- CALB Battery (25)
- Cylindrical Cell (9)
- Energy Storage System (0)
- Battery Management System (11)
- Sodium ion Battery Cell (4)
- Lithium Titanate Battery (22)
- Ternary Lithium Battery Cell (12)
- REPT Battery (10)
- BYD Battery (5)
- CATL Battery (15)
- Thunder Sky Winston Battery (26)
- EVE Battery (35)
- LiFePO4 Battery Cell (17)
Certification
Customer Reviews
- I have fond memories of our meeting in Shanghai with LiFePO4 Battery Shop Elena. Your company left a strong impression on me with its impressive growth and professionalism. We both value straightforwardness and honesty, which I believe are the most important qualities in any partnership. I am confident that we can build a successful collaboration based on these shared values. —— Robert from USA
- I've been working with LiFePO4 Battery Shop for years, and their reliability is unmatched. While other suppliers frequently change sales teams, LiFePO4 Battery Shop has consistently provided exceptional service with a stable team. Their commitment to quality and customer support truly sets them apart. —— Henry from Australia



